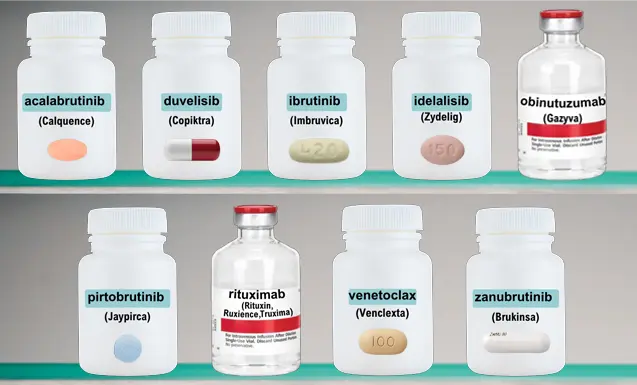
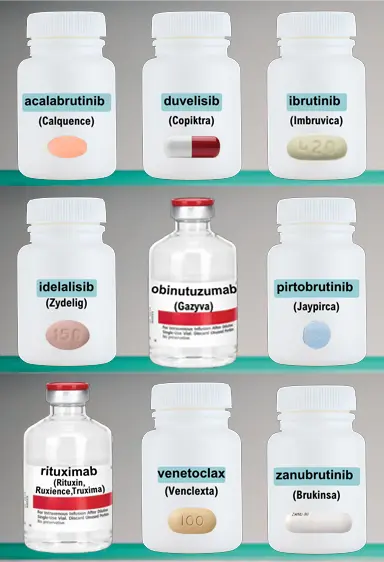
The CLL Medicine Cabinet
Choosing the Right Treatment for You
There are several factors your healthcare provider will take into consideration when discussing what treatment options might be the best for you. These factors may include:
- Your biomarker test results (which help determine staging, disease aggressiveness, and prognosis)
- Your comorbidities (other conditions that you already have in addition to cancer, such as heart or kidney disease)
- Your age
- Whether this is the first time you need treatment (first-line), or if this is a subsequent treatment (second-line and beyond)
- What the expected benefit will be from a treatment
- What the common side effects might be
- How the treatment is administered (oral, intravenously)
- Where the treatment will need to be administered (home, an infusion center, or a hospital)
- How long the treatment must be taken (for a limited duration of time, or continuously until the medication no longer controls the disease)
- Your financial considerations (will the medication be covered by your insurance and if not are there any ways to obtain the treatment through other means, such as medication assistance programs)
- Whether the treatment is already FDA-approved or if it only accessible through a clinical trial
Click on the individual bottles below to view a printable medication sheet and access links to other valuable information.