By Susan Leclair
DNA within a cell is made up of a ladder of two long strings of many deoxyribose molecules which can look like the two long poles of a ladder. The rungs of the ladder are compounds called nucleic acids. For those of you who knit, think of the deoxyribose as your knitting needles and the product of work that hangs between them as the nuclei acids. Groupings of these nucleic acids are called genes. Again, for the knitters, a gene could be the insertion of a cable stitch. When groupings are added together and active, they cause the cell to make a protein. Since proteins can be very complex, these structures can be very long. For example, one gene might have 500,000 of these nucleic acids.
Genes can make proteins that are found in essentially three large categories. The first is a protein that the cell needs in order to survive such as the enzymes necessary to use glucose to make energy.
Or they can make a protein which is placed on the outside of the cells and identifies the cell to other cells (Think wearing school colors at a football game) or they allow the cell to do something (Is the cell motile? Can it recognize foreign chemicals in the blood?)
Finally, these genes can make the cell produce compounds that are excreted into the blood stream. Think of antibodies which are made in one cell and then put out to float in the blood stream where they will act.
There are genes that are used only once in the life of a cell. While salamanders and other reptiles can regrow limbs, you cannot. What genes control limb development will not work ever again.
But there are other genes which must be used over and over again. It turns out that you make different hemoglobins at different times in your life. Fetal hemoglobin is made while you are a fetus and usually shuts down after you are born. Adult hemoglobin is made when you are a breathing human. But, oddly, when you need hemoglobin – say, as a result of a hemorrhage from a car accident, the genes that control the making of fetal hemoglobin turn back on, giving you two different methods of making hemoglobin which would allow you to recover from your blood loss faster. Then after you have a sufficient amount of hemoglobin, those fetal hemoglobin genes turn off again.
Still others can change their level of activity. All of us need Growth Hormone every day but none of us want to live through puberty more than once, which was bad enough. Certainly, the control of a woman’s menstrual cycle is radically changed when there is a pregnancy and all of those controls are under the oversight of genes turning on and off and regulating quantity.
How does this make any sense in lymphocytes? The processes are the same. The names are different. The most primitive cell is stimulated to become a lymphoid cell. This trigger is used once. The cell cannot change its mind and become a skin cell or a granulocyte. In the process of becoming mature, the cell has to turn on and off multiple genes to control growth in numbers. In other steps, the genes determine what functions are to be available at what steps. So, if the genes want a lymphocyte to be of a certain age before it can make antibodies. That means that this gene is not active until a stage in development. Again, for a silly analogy, lymphocytes start out as babies, so they have to learn to grow and recognize the world. Then as a child, they have to learn what their function is going to be. Then, they become teenagers when, as with human teenagers, they are supposed to develop the skills and attributes necessary for their adult lives. For example, getting a driver’s license. This stage of development for antibody producing lymphocytes is called pre-B because the totally functioning cells are B cells.
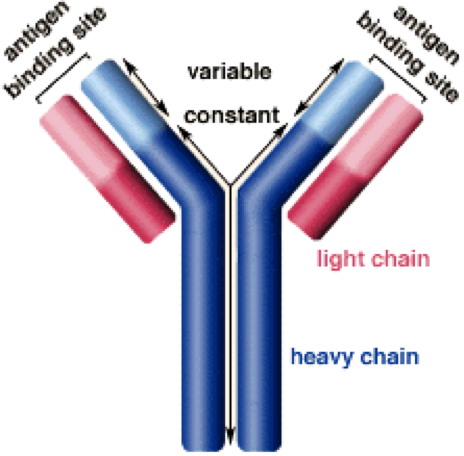
Antibodies look like two Y’s almost on top of each other.
To make an antibody, you need genes to control the making of the heavy chains, the light chains and the binding sites. As the graphic shows, the majority of the chains are the same even though you are making many different antibodies. It is the end region that makes an antibody specific to measles or mumps or a version of the flu virus.
Logic could assume that the cell has to grow up enough to first make all of the consistent or unchanging parts of the antibody before it can make those highly specialized regions. One of those genes controls the specialized portion of the upper arms of the heavy chain so it is unimaginatively named the Immunoglobulin Variable Heavy Chain gene or IgVH.
The IgVH genes exist in an inactive state until it is time to make antibodies. They then change from inactive to active or, in science lingo, they mutate. So, the non-mutated IgVH would be found in a younger cell when compared to the mutated or older cell.
One of the concepts about the world in general is that all structures start out as highly organized and orderly. As time and experience can afflict humans, so too do you find these consequences in cells. A cell is damaged and repaired, damage and repaired damaged and repaired until something goes wrong. That initial “wrongness” then puts additional stress on the cell so over time the cell begins to become less organized, less orderly. Just like the life of a human, most of us would prefer to have our diseases strike us when we are really really old rather than when we are twenty or ten years old. A cell that can still make one of its most important products – the antibody’s variable heavy chain part – is probably closer to the fully maturation stage than a cell that cannot make the variable heavy chain. So that would mean that the cell with the IgVH mutated (activated) gene is closer to functionally than the non-mutated gene. And that means that the mutated genes are in cells that have more defects to acquire over time than the younger non-mutated cells. Taking a longer time to reach a more serious state can translate into a longer watch and wait time or fewer complications or more response to some medications.
The world is not black and white in science either so notice the “fewer” or “longer”. This marker can only provide an approximation where the cell is in its downward spiral and give you a sense of longevity, but it is something and it is usable. And one more step to total control.
Susan Leclair is Chancellor Projessor Emerita at the University of Massachusetts Dartmouth; Senior Scientist, at Forensic DNA Associates; and Moderator and Speaker, PatientPower.info – an electronic resource for patients and health care providers.
Originally published in The CLL Tribune Q3 2018.