Ibrutinib and venetoclax are both highly effective treatments for chronic lymphocytic leukemia (CLL) that work through different mechanisms of action. Phase II clinical trials have shown that combining these two drugs is a very effective first-line treatment for patients who have not been previously treated for CLL, and it produces deep responses (meaning a large number of cancer cells are eliminated by the treatment). However, it is unknown whether there is any benefit to continuing treatment after patients reach undetectable minimal residual disease (uMRD) status. For an explanation of MRD testing, please see our MRD glossary or these resources.
At the annual meeting of the American Society of Hematology (ASH) 2020, our own Dr. Brian Koffman interviewed Dr. William Wierda, an Associate Professor of medicine in the Department of Leukemia at MD Anderson Cancer Center in Houston, Texas, about recent results from a clinical trial testing the combination of ibrutinib + venetoclax and MRD-guided discontinuation.
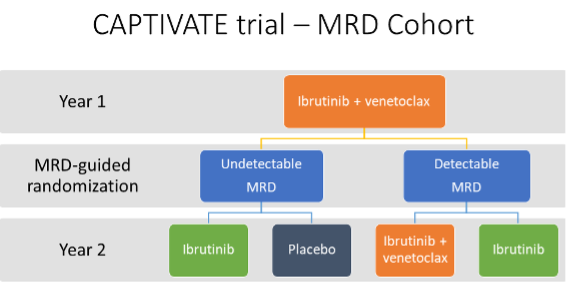
These are results from the CAPTIVATE study, a phase II clinical trial of ibrutinib + venetoclax for patients who had not previously been treated for CLL. The study design is summarized in the figure below. All patients received 12 months of treatment with ibrutinib + venetoclax, after which they were evaluated for MRD status in the bone marrow and blood. One-hundred forty-nine patients were then randomized to receive further treatment or placebo based on whether they had undetectable or detectable MRD.
Takeaways:
- Current novel CLL treatments fall into 2 categories:
- Continuous (ibrutinib, acalabrutinib) – must be taken continuously; good treatments but remissions aren’t usually deep enough to stop treatment
- Fixed-duration (venetoclax) – only used for a set, defined period of time; once patients get into a deep remission treatment is stopped
- After 1 year of treatment with ibrutinib + venetoclax, 86 patients had undetectable MRD and were randomized to ibrutinib only treatment or placebo.
- In undetectable MRD patients, there was no difference in 1-year disease-free survival between placebo and ibrutinib.
- These results indicate that there is no benefit to continuing treatment in patients who have undetectable MRD.
- For the 63 patients who had detectable MRD at randomization, follow-up is still ongoing.
- The MRD level of sensitivity for this study was 10-4 (1 in 10,000; flow cytometry), but researchers are moving towards using 10-6 (1 in 1,000,000; next-generation sequencing) as an indicator of the depth of response.
- Eventually clinicians would like to use MRD in community practices, and looking at MRD in the blood is probably a more practical approach than measuring it in bone marrow.
Conclusions:
The results from the CAPTIVATE trial demonstrate that fixed-duration ibrutinib + venetoclax is an effective treatment. For patients who achieve undetectable MRD, continuing treatment does not improve outcomes, which supports the idea of fixed-duration treatments followed by periods of no treatment. Researchers are moving towards treatment decisions that are MRD-guided (ie, treat until reach undetectable MRD rather than based on a fixed time), but clinical trial data is still needed to support this approach.
Please enjoy this interview with Dr. Wierda from the virtual ASH meeting which was held December 2020.
You can read the actual ASH abstract here: Ibrutinib (Ibr) Plus Venetoclax (Ven) for First-Line Treatment of Chronic Lymphocytic Leukemia (CLL)/Small Lymphocytic Lymphoma (SLL): 1-Year Disease-Free Survival (DFS) Results From the MRD Cohort of the Phase 2 CAPTIVATE Study
Take care of yourself first.
Ann Liu, PhD