Timothy Smith and colleagues presented this research at the American Society for Hematology annual meeting held in December 2021 (ASH 2021).
Background:
In recent years, the treatment paradigm of chronic lymphocytic leukemia (CLL) has changed significantly with the introduction of targeted therapies, which are substances that target specific enzymes, proteins, or other molecules involved in the growth and spread of cancer cells. For example, the Bruton tyrosine kinase inhibitor (BTKi) ibrutinib was approved for treating CLL in 2014, and the B-cell lymphoma 2 inhibitor (BCL2i) was approved in 2018. These therapies have led to improved outcomes and durable remissions for many patients. With more targeted therapies entering the clinic, researchers wanted to understand how CLL treatment patterns have changed over time in real-world clinical practices.
Takeaways:
- This was a retrospective analysis of two different real-world evidence databases.
- One data set came from an administrative claims database, which has information on diagnoses and procedures performed at each visit used for insurance reimbursement.
- The other data came from oncology practices’ electronic health records.
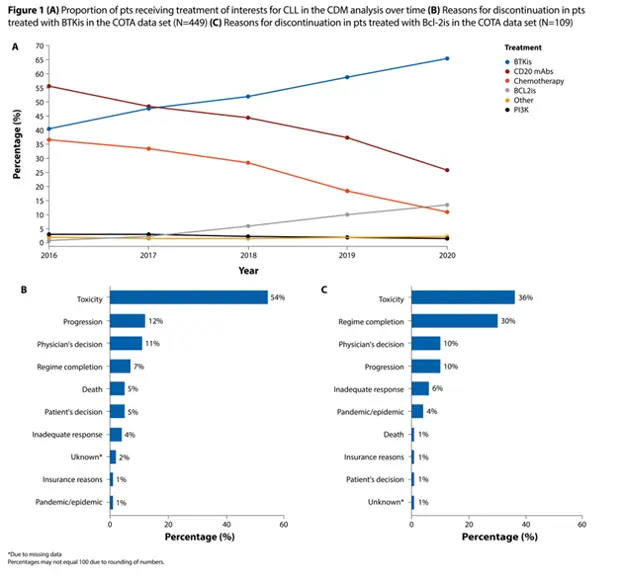
- Between 2016 and 2020, the proportion of patients treated with BTKis increased from 40% to 65%, and the proportion of patients treated with BCL2i increased from <1% to 13%.
- These increases were accompanied by corresponding decreases in chemotherapies and CD20 monoclonal antibodies.
- Most patients initiating BTKis were not previously treated (65%).
- In patients initiating BCL2is, most had received prior treatment (85%).
- More patients started on BTKis as first-line therapy (41%) compared with BCL2is (12%).
- Most patients who started targeted therapy as first-line therapy (81%) initiated BTKi monotherapy.
- Discontinuation rates were high for patients receiving BTKis (62%) and BCL2is (61%).
- Among BTKi discontinuers, toxicity (54%) was the most common reason for discontinuation, followed by disease progression (12%).
- Among BCL2i discontinuers, toxicity (36%) was the most common reason for discontinuation, followed by completion of the regimen (30%).
Conclusions:
This study showed that within a relatively short period of time standard of care shifted from chemoimmunotherapy to targeted therapies. However, patients receiving first-generation targeted therapies had high discontinuation rates, most commonly due to toxicities. Second-generation BTKi, such as acalabrutinib and zanubrutinib, are now coming into use. They are just as effective and have fewer side effects. Second-generation BCL2i are also in development. Science has brought CLL treatment a long way, and researchers continue developing new therapies and treatment regimens to improve patient outcomes.
Here is a figure from the abstract that illustrates some of the findings. Clearly, the move away from chemoimmunotherapy to targeted therapies is a positive trend for patients.
Here is the link to the ASH 2021 abstract for many more details.
Take care of yourself first.
Ann Liu, PhD

