Before getting into the numbers this week, we want to briefly touch upon the subject of Evusheld. Many in the CLL / SLL community have been asking CLL Society why the FDA has not yet made the decision to pull the emergency use authorization (EUA) for Evusheld, just as they did for Bebtelovimab several weeks ago when it no longer neutralized the majority of circulating variants in the United States.
After speaking with several experts on this subject, our understanding is that the FDA’s stance on Evusheld remains that while it no longer neutralizes the vast majority of circulating variants in the US (84.9%), there has been hesitancy to pull its EUA because Evusheld is the only COVID-19 monoclonal antibody preventative therapy available to at-risk immunocompromised individuals. For example, the thought is that as long as Evusheld retains activity against any remaining portion of the circulating variants, at least this is something versus nothing. This reasoning contrasts with their decision to pause the EUA for Bebtelovimab (and other previous COVID-19 monoclonal antibody therapies used for treatment that have lost their effectiveness), because there are still other COVID-19 treatment options remaining (such as the antivirals Paxlovid, Molnupiravir, and Remdesivir) in its absence. There are no other preventative options besides Evusheld.
CLL Society’s main concern with this approach is that there are many immunocompromised individuals who have received Evusheld that have no idea of its drastically reduced ability to neutralize the virus, so they are not taking additional necessary measures to protect themselves from infection right now. This is one of the reasons CLL Society began creating these weekly updates, so those in our community could perform more accurate personal risk assessments as the holiday season approaches, and to ultimately assist everyone in making fully informed decisions.
Weekly COVID-19 Statistics
Here is the CDC’s map of community level of spread (this map’s risk is weighted based on hospitalizations), and here is the link to the New York Times actual cases per capita map as shown below.
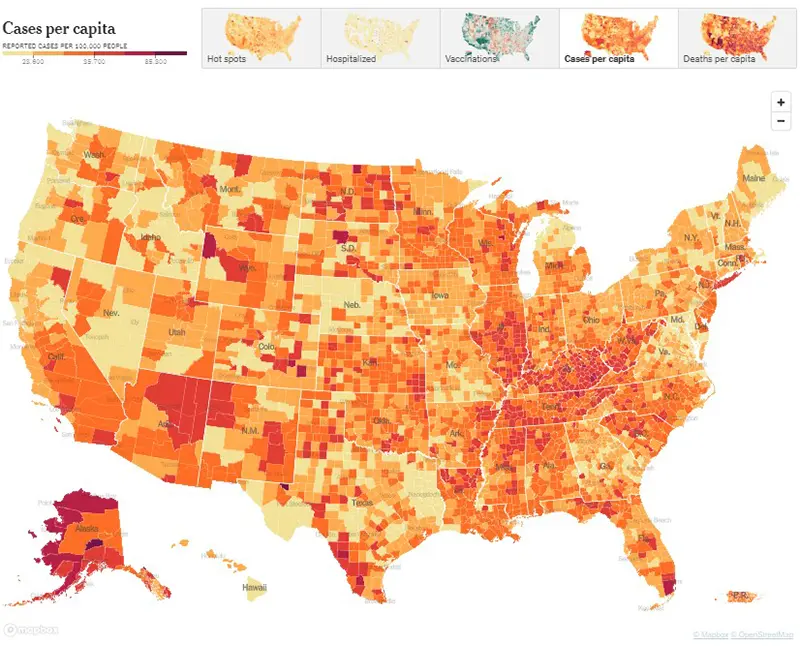
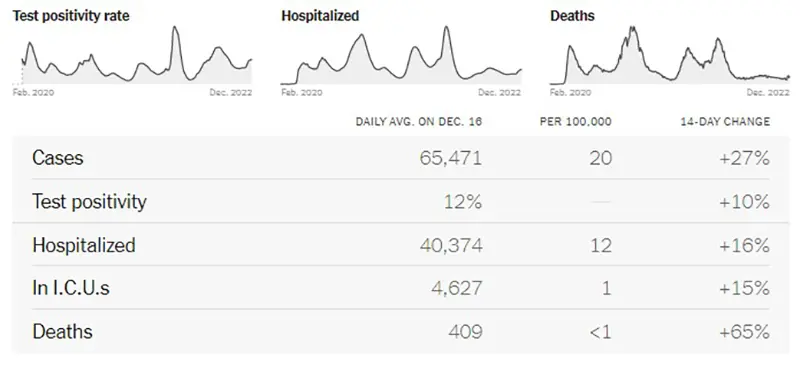
This graphic also from the New York Times says is very telling. All data points have increased significantly except for deaths, which is a lagging indicator behind cases and hospitalizations typically by about two weeks.
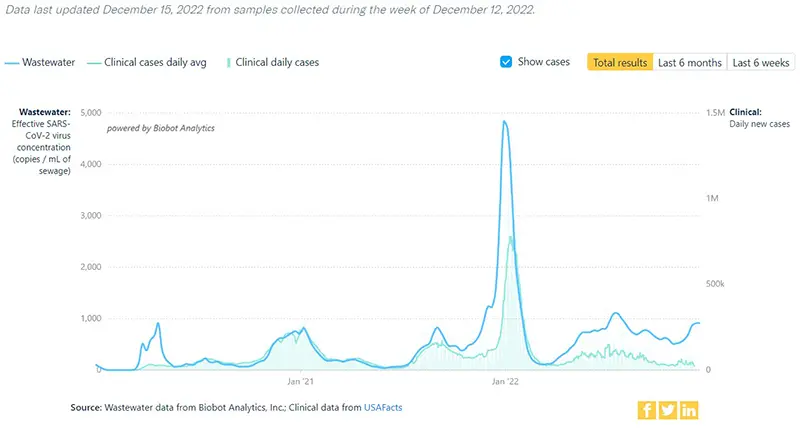
The COVID-19 Wastewater Monitoring Project last updated December 15th also indicates a sharp increase. Again, note that there is a large gap between the upper dark blue line (wastewater measurements of virus levels) and the lower light blue line (reported case numbers). Again, this map proves that actual case numbers are much higher than what is being reported due to either people not testing or only performing at-home tests.
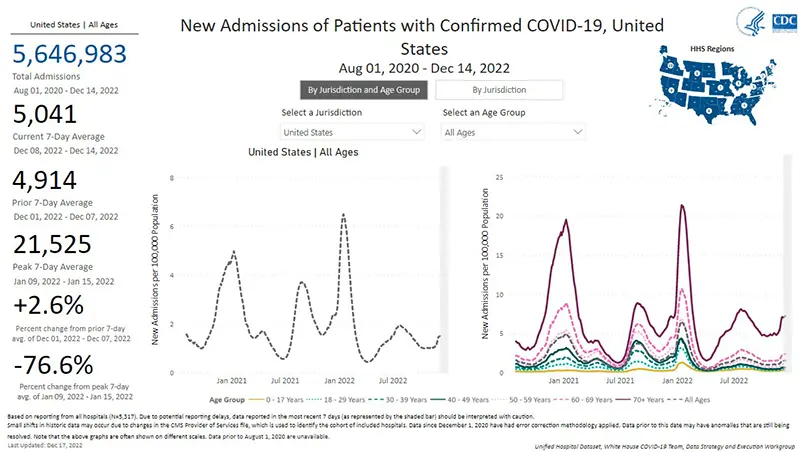
The rate of hospitalizations last updated on December 14th indicate hospitalizations for COVID-19 have gone up in all age groups. But perhaps the most concerning is the sharp contrast in hospitalizations for those over the age of 70.
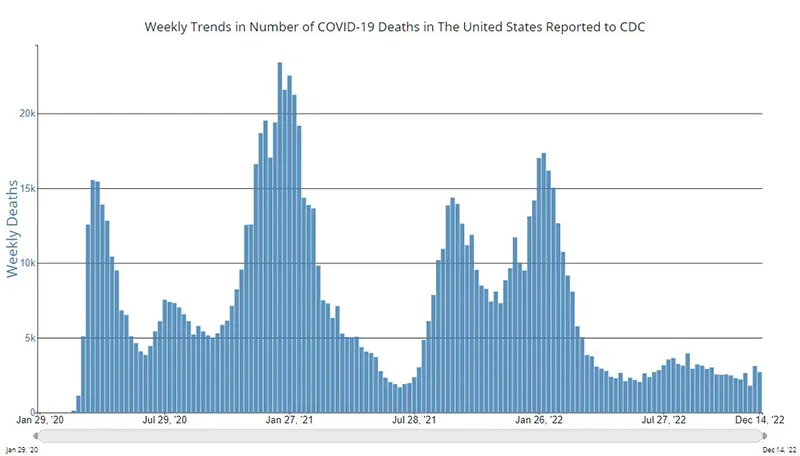
The weekly average number of COVID-19 deaths in the US ending the week of December 14th were 2,703. This is a decrease of 278 from the week prior.
Current & Emerging Variants of Concern (VOC)
BQ.1 and BQ.1.1 combined are now 69.1% of new cases this past week. Here is the overall breakdown of variants in the US according to the CDC’s Variant Tracker as of December 17th.
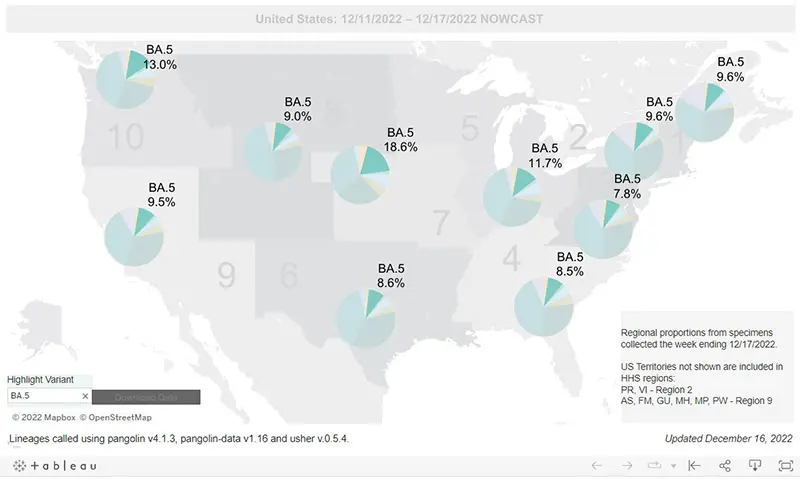
And here is the breakdown by regions within the United States as of December 17th. (We continue to highlight how much BA.5 is left by region because it is the main one that Evusheld still works against).
Here is what we know about the current variants of concern and how they hold up against Evusheld:
Data indicate Evusheld will still provide protection against the following variants (percentages are national averages):
- BA.5 (10.0%)
- BN.1 (4.1%)
- BA.2 (0.6%)
- BA.2.75 (0.5%)
These four variants together make up ~15.2% of cases on average nationwide as of December 17th.
Data indicate that Evusheld will NOT be effective against the following variants (percentages are national averages):
- BQ.1.1 (38.4%)
- BQ.1 (30.7%)
- XBB (7.2%)
- BF.7 (4.9%)
- BA.5.2.6 (1.6%)
- BA.4.6 (1.1%)
- BF.11 (0.7%)
- BA.2.75.2 (0.3%)
These seven variants together made up ~84.9% of cases on average nationwide as of December 17th.
Please discuss any questions you might have surrounding Evusheld with your healthcare provider.
COVID-19 in the News
- The White House announced last week that each household is now eligible to order four more free at-home COVID-19 rapid antigen tests though COVIDtests.gov as part of a new “winter preparedness plan” as cases and hospitalizations continue rising across the country. While it is important for those who are immunocompromised to have rapid antigen home tests on-hand, remember that these rapid antigen tests do not pick up an infection very well before or shortly after symptoms start, and they are known to be less accurate than PCR tests. However, rapid antigen tests are a good tool to use after an infection has been confirmed to help determine if there is still shedding of the virus which would mean the person is still considered infectious. Individuals are no longer infectious after having two negative rapid antigen tests that are taken 24 hours apart. If you have known exposure to COVID-19 or if you are exhibiting symptoms, please obtain a PCR test as soon as possible, since it can take up to 48 hours to receive PCR test results. PCR tests are more sensitive than rapid antigen tests because they detect the DNA of the virus instead proteins produced by the virus as it replicates. Of note, rapid home PCR tests have just become available to the public through Amazon and other websites within the past month and are extremely accurate. Most health insurance plans will reimburse individuals for the purchase of up to eight COVID-19 home tests per month.
- The CDC released a report on the effectiveness of the bivalent COVID-19 vaccines in preventing hospitalization among those 65 and older who are immunocompetent. Data indicate very high levels of vaccine effectiveness compared with either no vaccination or the original (monovalent) booster. The good news is that the bivalent booster against the current circulating variants provided up to 84% protection for the age group that is currently seeing the highest rates of hospitalization (those who are 65 and older). Unfortunately, right now we are seeing what some are referring to as the “senior COVID-19 wave” since hospitalizations among this age group are higher than what has ever been seen before in any other prior COVID-19 wave of the pandemic! As of the time this was written, only 36% of American seniors have received a bivalent booster within the last four months (we know immunity wanes by that 4-month mark). And while we need more data that are specific to the bivalent and the immunocompromised community, there was information presented at ASH demonstrating the importance of staying up-to-date on boosters, regardless of previously recorded antibody responses. Boosters can make a huge difference!
- It was announced that sometime in 2023 the Department of Health and Human Services (HHS) will stop supplying and distributing COVID-19 treatments, mainly due to lack of continued funding. This means pharmacies will begin to privately purchase and bill for COVID-19 therapies in the same way they do for all other medications. Currently, vaccines and treatments are available under an emergency use authorization from the FDA. It is expected the Paxlovid will be commercialized by mid-2023, and it is expected that the government will stop paying for COVID-19 vaccines sometime next year. CLL Society’s healthcare policy and advocacy team will continue working behind the scenes requesting the federal government ensures that the commercialization of COVID-19 vaccines and therapies, as well as the future lifting of the public health emergency, does not create unnecessary barriers to care for our at-risk immunocompromised community.
In Summary
- If you are eligible and have not yet received your COVID-19 bivalent booster, please consider obtaining it as soon as possible.
- Evusheld is only able to neutralize ~15% of the current circulating variants causing COVID-19 infections in the United States.
- If you have known exposure to COVID-19 or are experiencing any symptoms at all, please get tested early, preferably with a PCR test, and call your healthcare provider. Paxlovid must be started orally within five days of symptom onset and Remdesivir must be started intravenously within seven days of symptom onset.
- Wear a well-fitted N95 mask (or KN95) while around others who live outside of your household at all times. Surgical masks and cloth masks will not protect you if someone around you is infectious. You can obtain quality N95 masks free of charge from many local pharmacies across the country.
- Practice good hand washing often and use hand sanitizer.
- Practice social distancing and avoid large indoor gatherings or crowded situations as much as possible.
- Ensure there is good air-flow and ventilation whenever you are around others by opening windows or doors as weather allows, and/or using a quality HEPA air purifier if you have one available.
- Please revisit your COVID-19 Action Plan.
Keep learning, and please stay well.
Robyn Brumble, MSN, RN
Director of Scientific Affairs & Research
CLL Society



