On August 31, 2022, the FDA amended the emergency use authorizations (EUAs) for both the Moderna and Pfizer COVID-19 vaccines to include a bivalent formulation. The updated vaccine includes the same formula as the original vaccine but includes coverage against two additional strains of the coronavirus, BA.4 and BA.5. As of the time this article was written, BA.4, BA.4.6, and BA.5 (subvariants of Omicron) are the dominant circulating variants of concern in the US, and the newly updated bivalent vaccine does provide a stronger level of protection against them.
The CDC put out a press release on September 1, 2022, and then officially updated their recommended COVID-19 vaccine schedule for the moderately to severely immunocompromised on their website on September 2, 2022.
As a recap, if you have received either of the mRNA vaccines (Pfizer or Moderna), the “primary vaccine series” consists of three full-strength shots for CLL / SLL patients, NOT two (as is the case for those with a healthy immune system).
Then, no matter how many booster doses you have received previously in addition to the three primary doses in the series, the guidance has been simplified for everyone to include only the most recently approved COVID-19 bivalent as your only necessary booster dose. The bivalent booster should be received at least two months after your last dose of the vaccine (no matter if your last dose was a third primary series dose or a booster dose with the original version of the vaccine).
Having difficulty keeping it all straight? Here is a helpful graphic that was created by the CDC specifically for the immunocompromised to help keep everything straight:
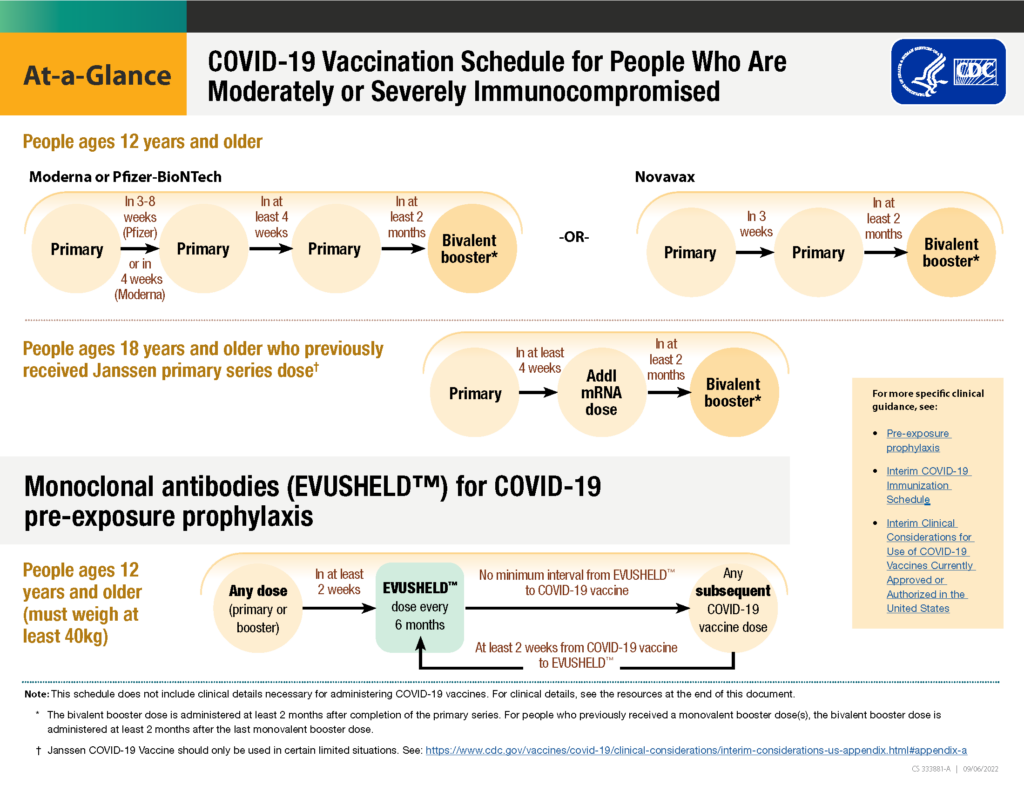
If you would like to view this graphic on the CDC website to view the links, you can do so here.
As a reminder, here are some important things to remember when it comes to COVID-19 vaccinations and CLL / SLL:
- Your COVID-19 antibody response to previous doses of the vaccine does not determine whether you should adhere to the CDC’s recommended COVID-19 vaccination schedule or not.
- Even if you have never had a robust antibody response to previous doses of the COVID-19 vaccines, you should still adhere to the recommended CDC schedule. This is because vaccines are known to stimulate other parts of our immune systems that are also important in fighting off the infection (such as T-cells, which are not at this time measurable with commonly available lab tests).
- All of those diagnosed with CLL / SLL should still receive Evusheld, regardless of their vaccination status, treatment status, or if they are still in Watch & Wait. But please remember that Evusheld is not to be used as a replacement for COVID-19 vaccinations.
- Additional COVID-19 vaccination doses are best if received before starting any new CLL / SLL treatment regimens since several common CLL / SLL therapies will diminish the body’s ability to respond adequately to vaccines.
- According to the CDC guidance, COVID-19 vaccines can be received either two weeks before Evusheld or any time after receiving Evusheld. Although, many CLL Experts still recommend ideally receiving the vaccine dose first, then obtaining Evusheld at least two weeks later when possible.
- Please make sure you are keeping your COVID-19 Action Plan current.
As has been the case since the start of the pandemic, the only thing we know for sure when it comes to COVID-19 is that it will continue to keep us guessing in real-time as new variants continue to emerge. So, it is important to keep in mind that eventually, these most recent set of recommendations will also change.
The one thing that has remained the same throughout the pandemic is that we must all stay educated about what is happening in the here and now, and continue to protect ourselves to the best of our ability with a multi-faceted approach to infection control (continue masking, avoiding large crowds especially indoors, and stay up-to-date with vaccinations and Evusheld dosing).
Keep learning and stay well.
Robyn Brumble, MSN, RN
Director of Scientific Affairs & Research
CLL Society

